Links from system analysis to mathematical modeling
The quantitative analysis of a biological system is not possible without first constructing a simple algorithmic model. A model can provide important insights into the structure of a system, and it does increase our understanding of the system itself. Modeling can also reveal the logical errors of simple biological and clinical concepts. Moreover, to be able to simulate the complete or incomplete failure of a single element or an entire pathway permits us to pose direct clinical questions in Neurology and Neurosurgery about the localization of the damage as well as the mechanism involved.
The vestibulo-ocular reflex can serve as an example. A basic model of the vestibulo-ocular reflex consists of measurable quantities of system input (head velocity), which influence system components that communicate with each other and are also directly and reciprocally coupled with each other. System output is again defined as measurable quantities (eye position). In a model in which the input is a head turn and the output eye movements, the vestibulo-ocular reflex drives the eyes to move in the opposite direction of the turning head. This keeps the gaze on the target stable. Static models describe the vestibular ocular reflex by means of mathematical matrices, whereas dynamic models use elements of linear system theory, such as high and low-pass filters. Another approach uses neural networks with non-linear features of model neurons and their connections. By means of such networks, the relationship between the system elements can be weighted according to their importance and adjusted to take learning processes into account.
This was accomplished, for example, in a basic version of a 3-dimensional mathematical sensorimotor feed-forward model that elucidated otolithic control of binocular static eye position. The mathematical model depicted in the figure below can accurately simulate 3-dimensional ocular deviations of patients who have unilateral utricular loss, complete vestibular nerve failure, or lesions of the vestibular nuclei or the pathways for graviception in the brainstem.
A saccadic burst generator and the neural velocity to eye position integrator, including the experimentally demonstrated leakage in the torsional component had to be implemented in the model as well as the otolith-dependent neural control of Listing's plane in order to simulate cerebellar and brainstem disorders with pathological nystagmus (Glasauer et al.2002).
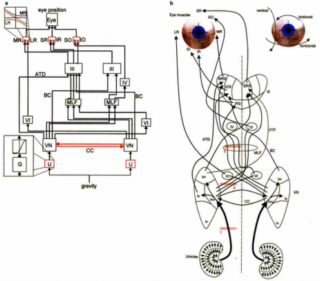 Figure left depicts the block diagram of the three-dimensional mathematical model of utricular-otolith control of the static eye position. Only the connections to the left eye are shown for the sake of clarity. Connections from the left utricle (U) via the vestibular nuclei (VN) through the medial longitudinal fasciculus (MLF), the brachium conjunctivum (BC), and the ascending tract of Deiters (ATD) via the ocular motor nuclei (III, IV, VI) to the extraocular muscles (agonists: medial rectus MR, superior rectus SR, superior oblique SO; antagonists: lateral rectus LR, inferior rectus IR, inferior oblique IO) of the left eye are shown in blue. Additions to the basic version of the model are shown in red: excitatory commissural connections (CC), the non-linear force-response relationship of utricular peripheral neurons (inset on the lower left; G, utricular geometry), and the quadratic innervation-force characteristic of extraocular eye muscles (inset, upper left). (b) A more anatomical presentation of the block diagram, showing the brainstem pathways from the utricles and the vestibular nuclei (sv, superior vestibular nuclei; mv, medial vestibular nuclei; lv, lateral vestibular nuclei; iv, inferior vestibular nuclei) via the ocular motor nuclei (MRS, medial rectus section; SRS, superior rectus section; IOS, inferior oblique section; IRS, inferior rectus section) to the extraocular eye muscles. 1, 2, and 3 indicate simulated lesions of the utricular nerve (1), medial vestibular nucleus efference (2), and ascending graviceptive pathways in the MLF (3).
Figure left depicts the block diagram of the three-dimensional mathematical model of utricular-otolith control of the static eye position. Only the connections to the left eye are shown for the sake of clarity. Connections from the left utricle (U) via the vestibular nuclei (VN) through the medial longitudinal fasciculus (MLF), the brachium conjunctivum (BC), and the ascending tract of Deiters (ATD) via the ocular motor nuclei (III, IV, VI) to the extraocular muscles (agonists: medial rectus MR, superior rectus SR, superior oblique SO; antagonists: lateral rectus LR, inferior rectus IR, inferior oblique IO) of the left eye are shown in blue. Additions to the basic version of the model are shown in red: excitatory commissural connections (CC), the non-linear force-response relationship of utricular peripheral neurons (inset on the lower left; G, utricular geometry), and the quadratic innervation-force characteristic of extraocular eye muscles (inset, upper left). (b) A more anatomical presentation of the block diagram, showing the brainstem pathways from the utricles and the vestibular nuclei (sv, superior vestibular nuclei; mv, medial vestibular nuclei; lv, lateral vestibular nuclei; iv, inferior vestibular nuclei) via the ocular motor nuclei (MRS, medial rectus section; SRS, superior rectus section; IOS, inferior oblique section; IRS, inferior rectus section) to the extraocular eye muscles. 1, 2, and 3 indicate simulated lesions of the utricular nerve (1), medial vestibular nucleus efference (2), and ascending graviceptive pathways in the MLF (3).





